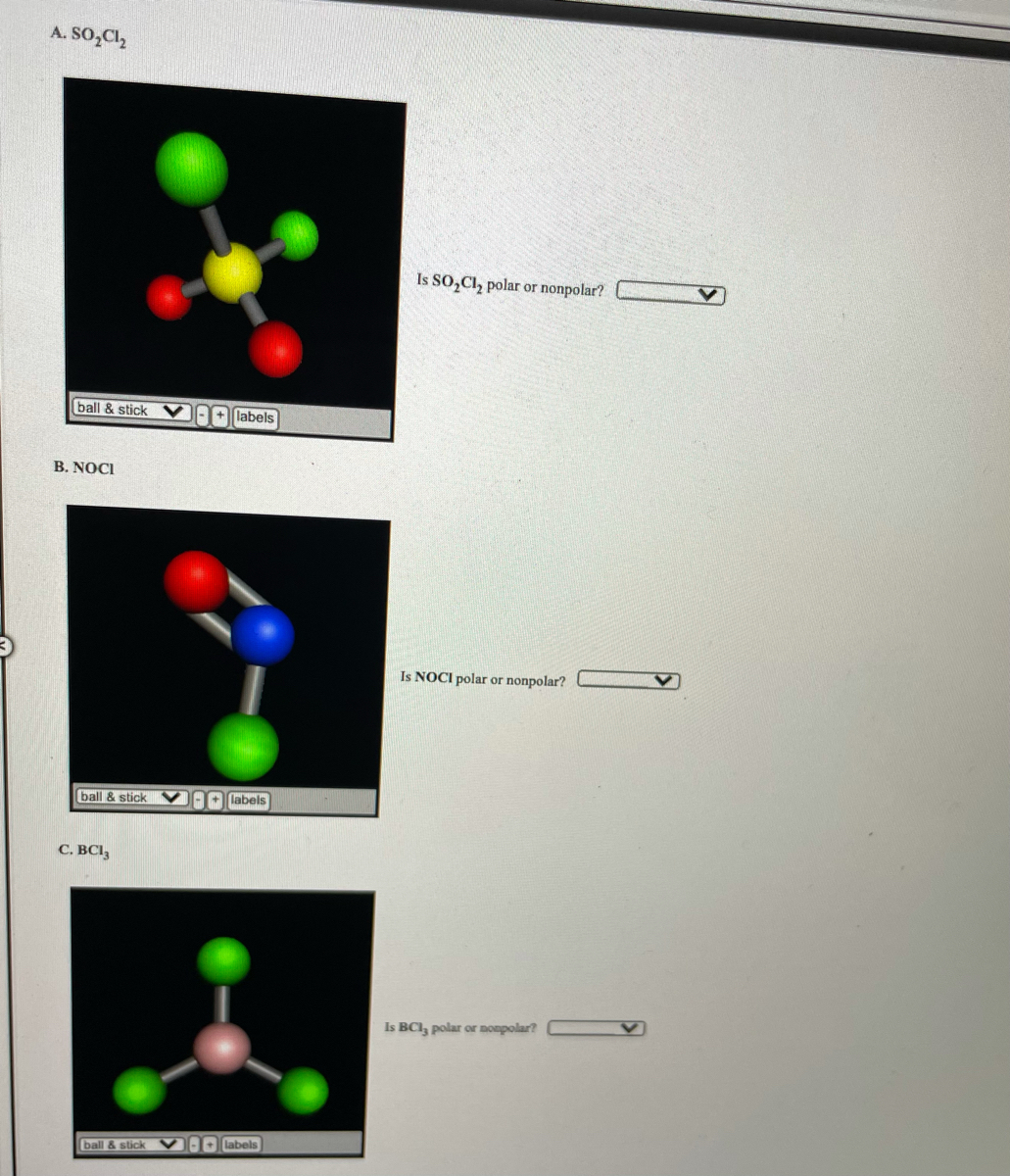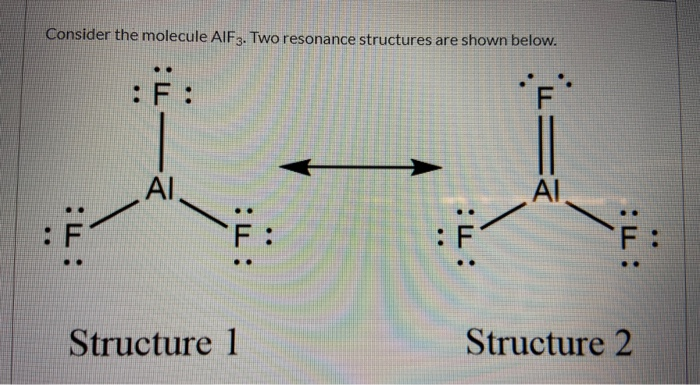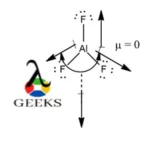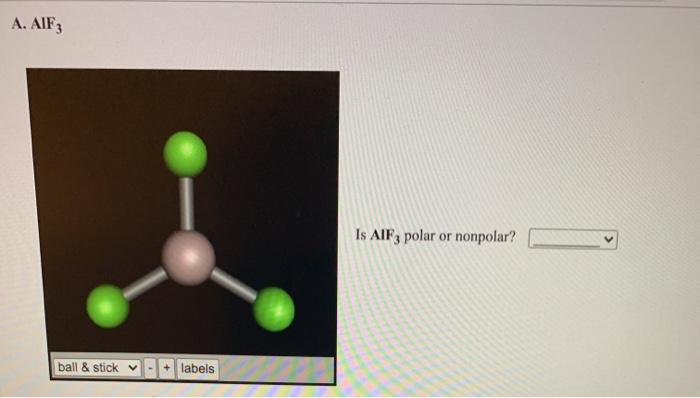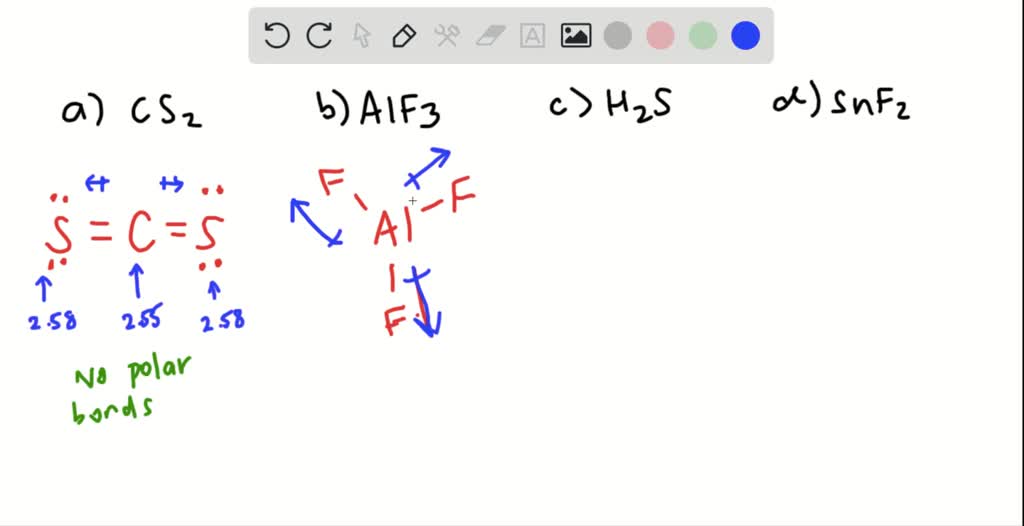
SOLVED:Write the Lewis formula for each of the following. Indicate which bonds are polar. (See Table 5-3 . ) Indicate which molecules are polar. (a) CS2 ; (b) AlF3 ; (c) H2 S ; (d) SnF2
Both BF3 and NF3 are covalent compounds but NF3 is a polar compound while BF3 is non-polar. How can you explain it? - Quora
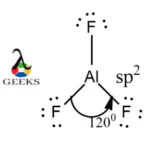
Shapes of Molecules – Honors Chemistry I Study Guide H2S: 1+1+6=8 AsCl3 AlF3(Note: aluminum has an incomplete octet in this