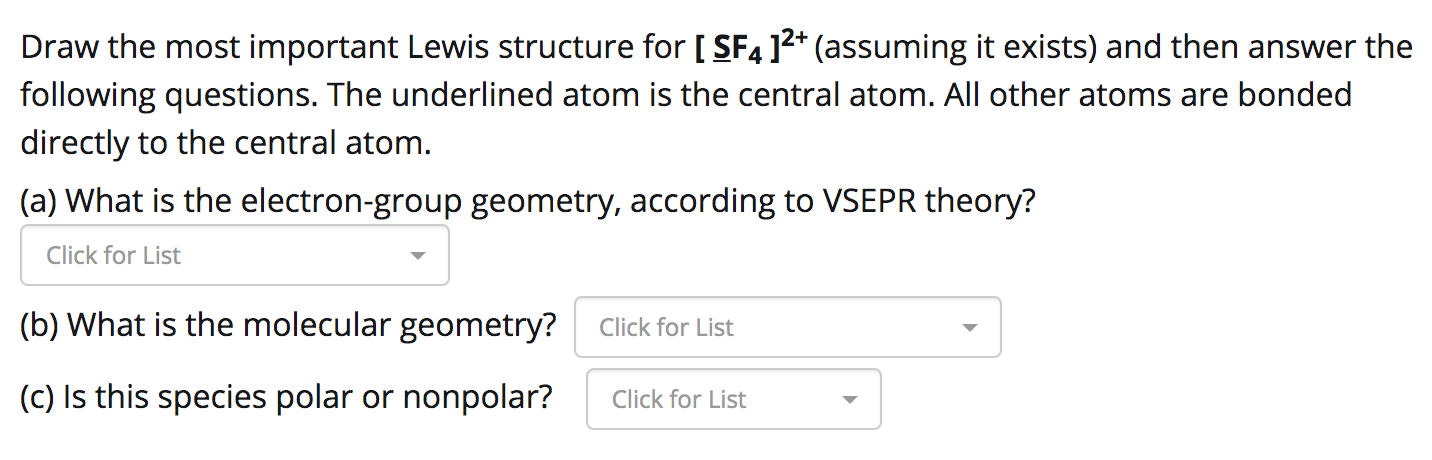
Your Turn! A central atom has two lone pair of electrons around it and two single bonds to other atoms. What is the electron pair geometry around the central. - ppt download

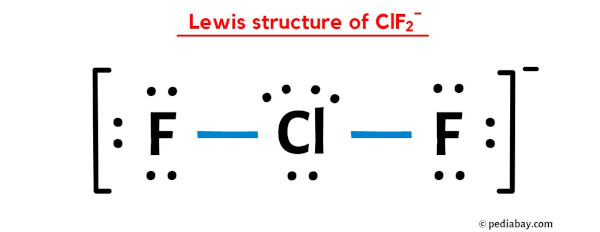
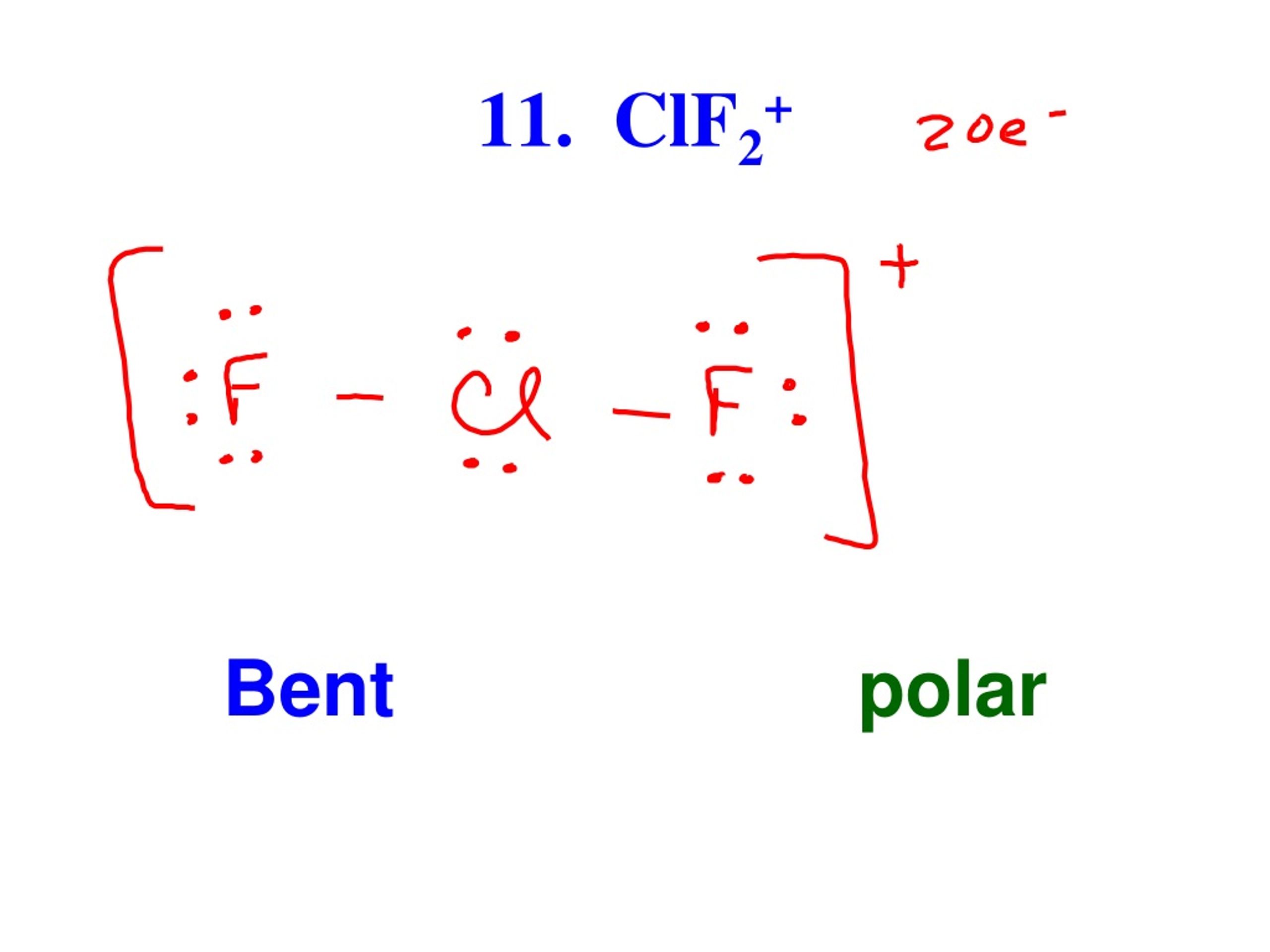
_____Good evening _____Answer this:-Explain why CIF2- is linear but CIF2+ is bent molecular ion. - Brainly.in

Is ClF Polar or Nonpolar (Chlorine Monoluoride) | Is ClF Polar or Nonpolar (Chlorine Monoluoride) Hello Everyone! Welcome back to our channel, and for today's video, we will help you determine if...

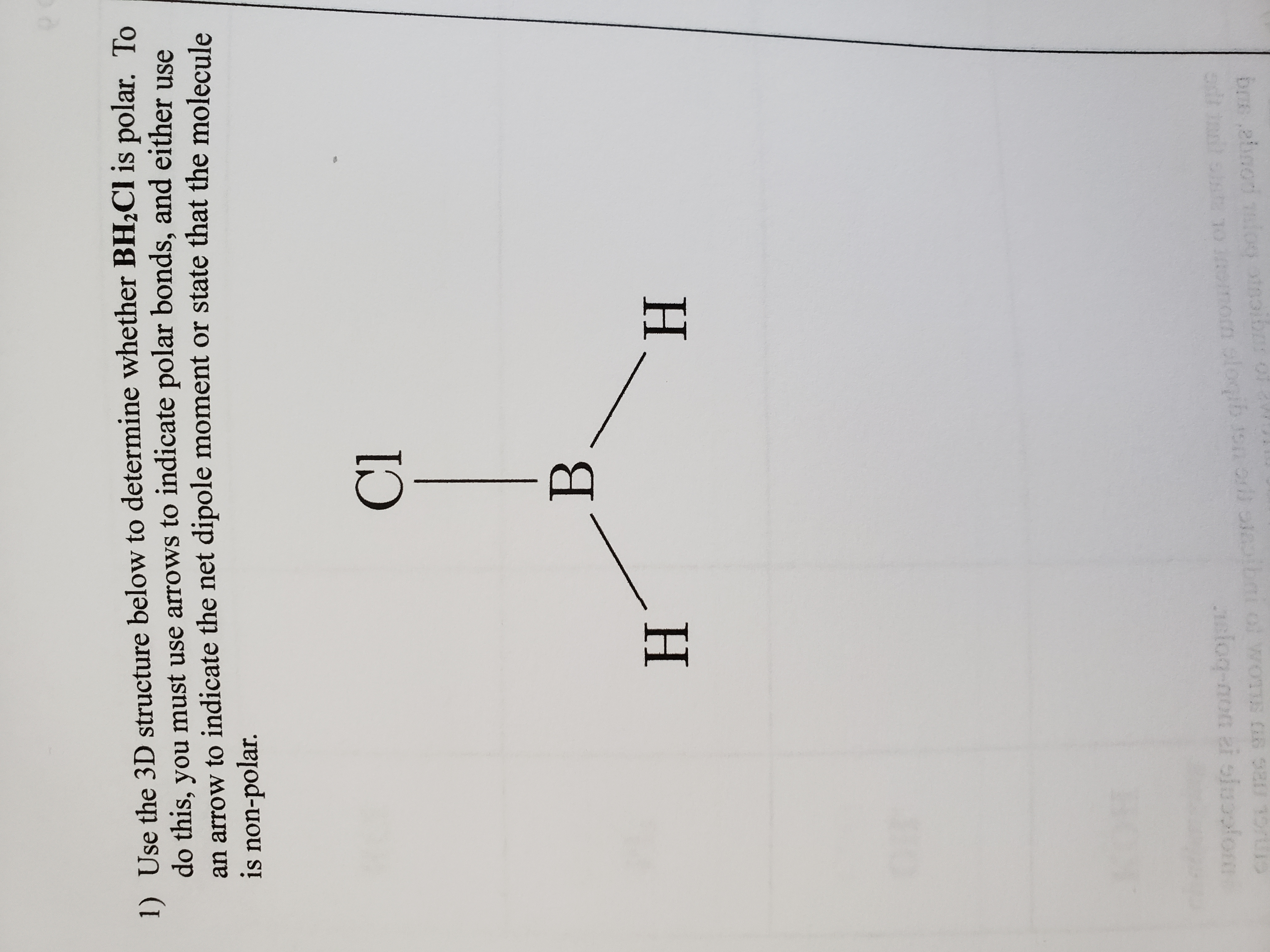
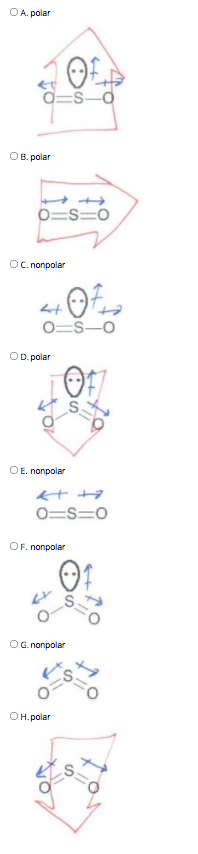
PS 18 2019_Su2021.docx - Problem Set 18 – Molecular Shape, Polarity Chem 105 1. Once you know the concepts of bond dipoles and molecular shape, you can | Course Hero